|
7/14/2023 0 Comments Covid vaccination card hipaa 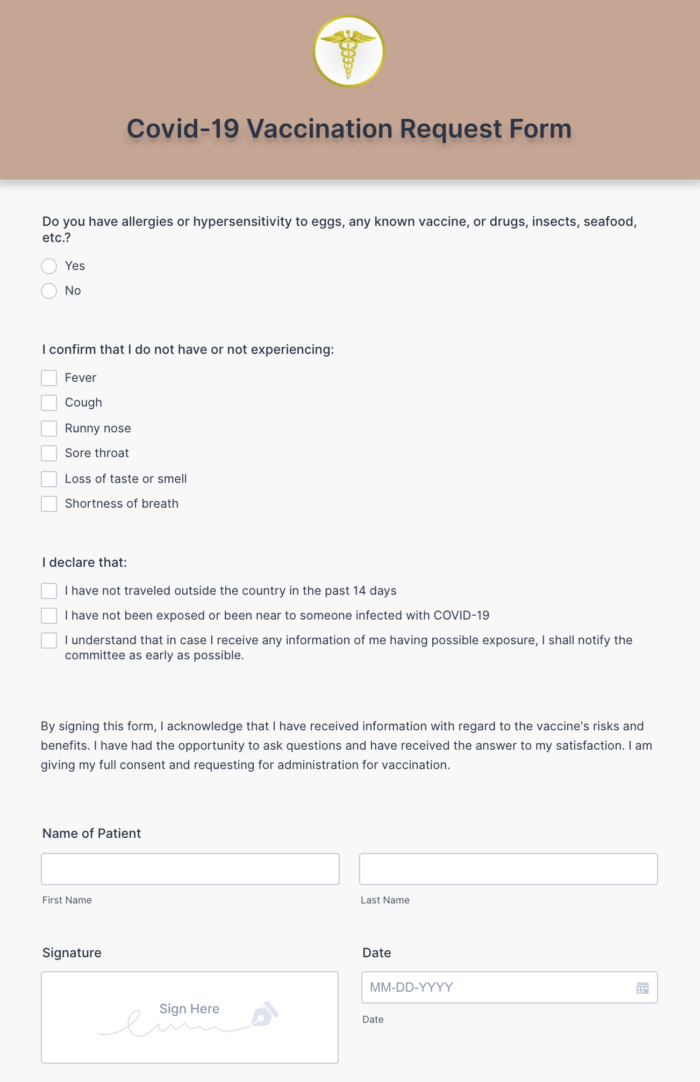 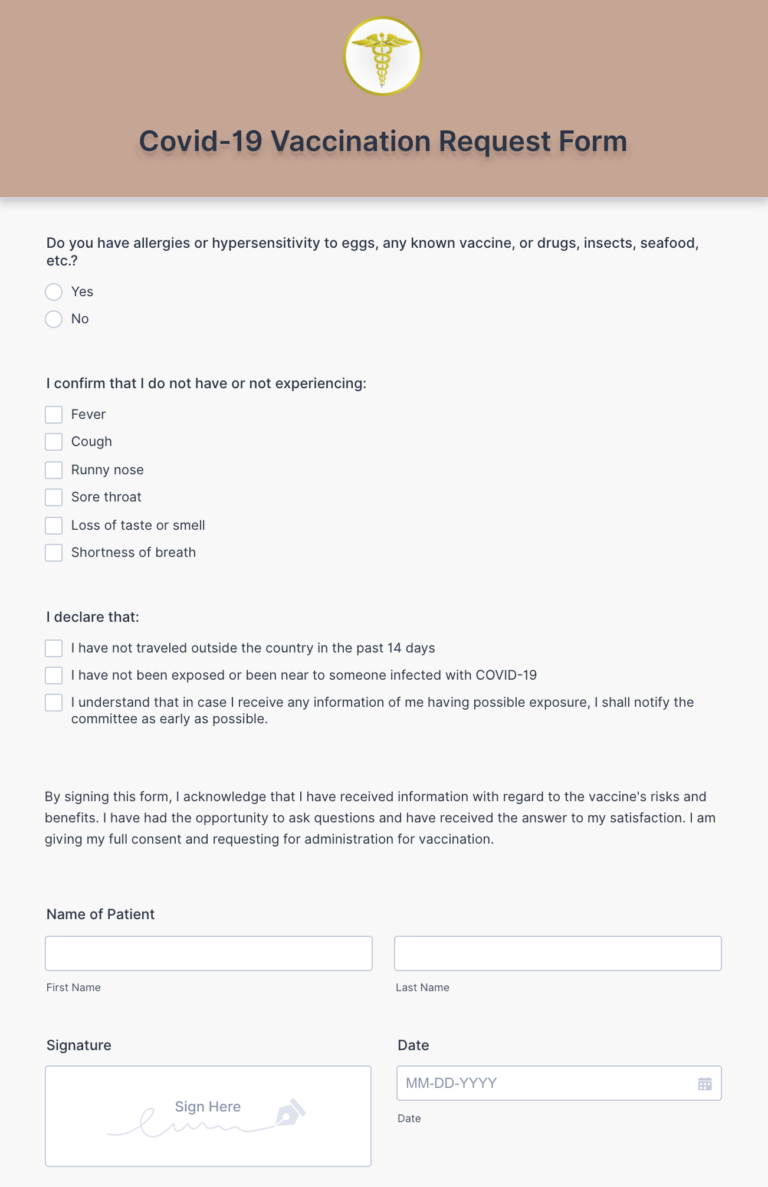
In the UK, mRNA Moderna and Pfizer-BioNTech vaccines have been approved for use in seasonal booster programs. T he UK government is not ordering future supplies of the AstraZeneca COVID-19 vaccine, according to the British Heart Foundation. The UK became the first country in the world to approve the Oxford University/AstraZeneca COVID-19 vaccine in 2020. 
“Strict time limits apply to such claims so anyone who thinks they may have been affected in a similar way should get in touch,” he added. Todd previously told The Epoch Times that the UK government has “agreed in the procurement contract of the vaccine to indemnify AstraZeneca against any liability of this kind, so we ask the government to do the right thing and to support people who were vaccinated and help them cope with the devastating consequence of their life-changing injuries.” “I don’t think they are yet accepting they are causing other injuries, hence why they have made no awards,” he added. “I think the government accepts the mRNA vaccines are causing myocarditis/pericarditis but I think they are trying to suggest the prognosis for those conditions is generally good,” he said. The Epoch Times asked Todd why the AstraZeneca vaccine is the only one that is damaging above the 60 percent threshold rule. The legal team is seeking personal injury compensation from AstraZeneca, claiming that the vaccine was “not as safe as the public were entitled to expect.” Peter Todd, a consultant solicitor with Scott-Moncrieff & Associates, has initiated a claim against AstraZeneca under the Consumer Protection Act 1987, representing dozens of people who say they suffered severe reactions or family members died after taking the vaccine. Caroline Nicolls receives an injection of the Moderna COVID-19 vaccine administered by nurse Amy Nash, at the Madejski Stadium in Reading, England, on April 13, 2021. Of these 133 claims, the number broken down by vaccine type was 92 for AstraZeneca, 31 for Pfizer, and 10 for Moderna. The government’s high bar for acceptance requires that all eligible applicants in the UK must meet a strict 60 percent disablement criteria. The publication claims that 105 claims have now been approved, taking the total bill so far to £12.6 million.Īccording to the FOI, 133 claims were unsuccessful because, although the claim met the criteria for causation, the independent medical assessor recommended that the vaccine has “not caused severe disablement.” The Daily Mail reported in June that the COVID-19 Vaccine Damage All-Party Parliamentary Group was told by health minister Maria Caulfield that, to date, 5,738 applications had been submitted to the scheme, up from 5,708 in May. Claimsīut the latest numbers could even be higher. Victims who got paid have predominantly developed vaccine-induced immune thrombocytopenia and thrombosis/cerebral venous sinus thrombosis and Guillain-Barre syndrome. This is up from March when 44 claimants were notified they were entitled to payment. All of the claims were awarded where the AstraZeneca vaccine was used. In December 2020, ministers agreed to add COVID-19 to the scheme to demonstrate “government confidence in the safety profile” of any vaccine being used in the vaccination programme.Ĭlaims are handled by the National Health Service Business Services Authority, an arm’s length body of the Department of Health and Social Care.Īccording to an FOI dated May 22, 96 claimants have been notified they are entitled to a Vaccine Damage Payment. Under current rules, victims of COVID-19 vaccines in the UK are entitled to a one-off sum of £120,000 under the Vaccine Damage Payment Scheme, if someone can prove they have been severely disabled or have had family die as a result of vaccination. UK government payouts for the vaccine injured have now surpassed £11.5 million but could be even more. Data obtained through Freedom of Information (FOI) requests have revealed a rise in successful COVID-19 vaccine injury claims which has nearly doubled in three months. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
